
Introduction
In banking, healthcare, insurance, and telecom, a poorly governed knowledge base isn't just an operational headache — it's a compliance liability. HHS has collected $144.9 million across 152 HIPAA settlements since 2003, and GDPR fines reached approximately €1.2 billion in 2024 alone. Outdated or ungoverned knowledge base content sits at the center of those failures — driving agent errors, regulatory violations, and audit findings.
Content governance — in this context — means defined ownership, audit trails, mandatory review cycles, and role-based access tied to your compliance obligations. In regulated industries, knowledge base content faces the same scrutiny as customer-facing disclosures, clinical protocols, or financial product materials. A style guide won't cover that.
This article lays out a practical framework for keeping your knowledge base compliant — covering content audits, ownership structures, review triggers, and the governance gaps that most often lead to regulatory exposure.
TLDR
- Content governance requires named ownership, audit trails, review cycles, and role-based access — not just editorial standards
- HIPAA, GDPR, FCA, and FINRA mandates make informal content management a direct regulatory liability in healthcare, banking, insurance, and telecom
- The most common failures: unclear ownership, missing version control, and skipped reviews
- Built-in compliance certifications (GDPR, SOC 2, ISO 27001, HIPAA) are a non-negotiable requirement when evaluating knowledge management platforms
How to Build a Compliant Knowledge Base Governance Framework
Step 1: Audit Your Existing Knowledge Base for Compliance Gaps
You can't govern what you haven't mapped. Begin with a complete inventory of all knowledge base assets — articles, decision trees, SOPs, FAQs, visual guides — and flag items that lack ownership, review dates, or compliance validation.
What to look for:
- Content with no documented last-reviewed timestamp
- Articles with no named owner or designated approver
- References to outdated regulatory frameworks or retired policies
- Items that support teams have flagged as confusing or contradictory
Regulatory context: The FCA Handbook PRIN 2.1 (Principle 7) requires firms to "pay due regard to the information needs of clients, and communicate information to them in a way which is clear, fair and not misleading." Since July 2023, FCA Principle 12 (Consumer Duty) goes further, requiring firms to prove their communications support customer understanding — not merely avoid misleading. This directly implicates knowledge base content quality.
FINRA Rule 2210 is equally explicit: no member may publish any communication containing "any untrue statement of material fact or is otherwise false or misleading." The rule requires prior principal approval for retail communications, meaning KB articles supporting retail customer interactions may require documented review workflows.
The audit's goal: surface every article where regulatory alignment isn't documented or provable — before an auditor surfaces it first.
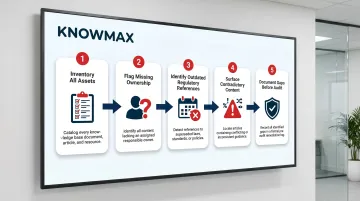
Step 2: Define Content Ownership and Approval Roles
Every knowledge base article in a regulated environment must have a named owner responsible for accuracy and a designated approver who validates content before publication. Without both, there's no chain of accountability when regulators ask who reviewed what — and when.
Ownership requirements:
- Content owner — responsible for accuracy, updates, and flagging when regulations change
- Approver — validates content meets regulatory and policy standards before publish
- Domain-specific expertise — healthcare content requires clinical or compliance sign-off; financial content needs legal or risk review
Domain expertise in practice:
Generic editorial approval doesn't hold up under scrutiny. HIPAA-governed patient data protocols need sign-off from someone with compliance authority in that clinical domain. KYC/AML procedures in banking require legal or risk team validation — not just a content editor.
Knowmax supports content ownership assignment at the article level, with a maker-checker approval process. Team leaders and compliance officers can review, suggest changes, and approve content before it goes live — ensuring alignment with regulatory standards.
Step 3: Establish Mandatory Review Cycles Tied to Regulatory Change
Unlike marketing content, regulated knowledge base articles can't be reviewed "whenever convenient." Review cycles should be tied to regulatory update calendars and risk tiers.
Dual-trigger review model:
ISO 27001:2022 Clause 7.5.3 mandates that documented information be reviewed at least every 12 months. Annex A Control 5.1 adds that policies must also be reviewed "when significant changes occur." Together, these create a dual-trigger model:
- Scheduled reviews at maximum 12-month intervals for all content
- Event-driven reviews triggered when regulations change, products launch, or processes shift
Tiered review schedule:
- High-risk content (compliance-sensitive procedures, legal disclosures, clinical protocols) — quarterly review
- General operational content (product FAQs, troubleshooting guides) — semi-annual or annual review
- All content — immediate review when applicable regulations are updated
Regulatory update velocity: FINRA alone issued at least 19 regulatory notices in 2025, plus weekly rule updates. The FCA publishes rolling handbook notices throughout the year. Organizations in financial services or healthcare need event-driven review triggers, not just annual schedules.
Step 4: Implement Version Control and Audit Trails
In regulated environments, version control serves as the evidence trail auditors require. Every edit, approval, and publish action must be logged with a timestamp and user identity — so you can reconstruct exactly what any agent saw, and when.
Regulatory requirements across frameworks:
| Framework | Requirement | What It Mandates |
|---|---|---|
| HIPAA Security Rule | Audit Controls (45 CFR 164.312(b)) | Hardware/software mechanisms to record and examine system activity |
| HIPAA Security Rule | Integrity Controls (45 CFR 164.312(c)) | Policies to protect ePHI from improper alteration or destruction |
| GDPR | Accountability Principle (Article 5(2)) | Controllers must demonstrate compliance with processing principles |
| ISO 27001:2022 | Document Control (Clause 7.5.3) | Full version history with who/what/when/why; named owners; archived versions |
| NIST SP 800-53 Rev. 5 | Change Control (CM-3) | Determine, document, and approve all system changes |
| SOC 2 | Change Management (CC8) | Documented change management processes |

What audit trails protect:
In the event of a customer complaint or regulatory investigation, audit trails prove agents were given accurate, current information at the time of interaction. This evidence can be the difference between a finding of willful neglect and a finding of reasonable cause — a penalty differential of $71,162 vs. $1,424 per violation under HIPAA.
Knowmax's platform logs every content change, approval action, and publish event with timestamped user attribution — giving compliance teams the audit-ready documentation they need under HIPAA, GDPR, and SOC 2 frameworks.
Step 5: Set Role-Based Access Controls for Sensitive Content
Not all agents or users should access all knowledge base content. In regulated industries, some information is restricted by law — patient data protocols under HIPAA, KYC/AML procedures in banking, or PII-handling workflows under GDPR.
Regulatory mandates:
HIPAA 45 CFR 164.312(a) requires technical policies to "allow access only to those persons or software programs that have been granted access rights," including unique user identification. This aligns with the Privacy Rule's "minimum necessary" standard for information access.
GDPR Article 32 requires "the ability to ensure the ongoing confidentiality, integrity, availability and resilience of processing systems," necessitating role-based access to systems containing personal data.
Access tier structure:
- View-only — frontline agents accessing approved content during customer interactions
- Edit access — content owners updating articles within their domain
- Publish access — compliance-approved reviewers who validate and release content
- Admin access — governance leads managing roles, permissions, and audit configurations
The cost of overly permissive access: The 2025 Ponemon Cost of Insider Risks Report found insider risk costs average $17.4 million per organization annually, with containment averaging 81 days. Incidents contained within 31 days cost $10.6 million; those taking 91+ days reach $18.7 million — nearly double. Tight access controls don't just limit exposure; they cut the time-to-detection that determines how much an incident ultimately costs.
Knowmax provides role-based access control (RBAC) that allows administrators to assign permissions based on departments, designations, or tasks. For example, healthcare organizations can restrict access to HIPAA-sensitive content to authorized personnel only, reducing data breach risk.
When Should You Revisit Your Knowledge Base Governance?
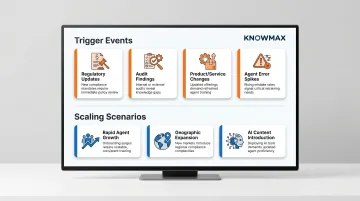
Governance isn't a one-time setup — it's a living system. Specific triggers signal when a governance review is needed:
Trigger events:
- New laws, updated guidance, or revised standards go into effect
- Audits surface content gaps, outdated articles, or missing approvals
- New offerings, retired services, or workflow updates make existing articles inaccurate
- Increased agent mistakes in a specific topic area point to underlying content problems
When governance models become insufficient:
- Rapid scaling — adding agents, content types, or new channels like chatbots and self-service portals — strains existing workflows
- Geographic expansion into new regulated markets with different compliance requirements, for example US healthcare operations extending to the EU under GDPR
- Introducing AI-generated content, which requires additional review and approval layers before publication
Organizations operating across multiple regulated sectors — for example, a BPO serving both healthcare and financial services clients — need governance frameworks that handle different compliance standards within a single environment. Knowmax's multi-tenant architecture lets each client operate under its own compliance controls, which matters when teams are managing HIPAA and financial regulations side by side.
What You Need Before Setting Up a Governance Framework
Equipment and System Requirements
A knowledge management platform capable of supporting regulated governance must include:
- Version control with full audit trails — logging every edit, approval, and publish action
- Role-based access controls — restricting editing and viewing by user role
- Content expiry and mandatory review alerts — flagging content that requires re-validation
- Compliance certifications — GDPR, SOC 2, ISO 27001, HIPAA
Certifications aren't marketing badges. They confirm the platform infrastructure meets baseline security and data governance requirements before you build a governance layer on top.
Knowmax holds GDPR, SOC 2, ISO 27001, and HIPAA certifications. SOC 2 Type II evaluates operating effectiveness over 6–12 months across access controls, change management, and monitoring. ISO 27001 requires version control with full history, role-based access, and 12-month review cycles — all capabilities built into the platform.
Inputs, Stakeholders, and Compliance Documentation
Before operationalizing governance, ensure you have:
- Current regulatory requirements document for your industry (HIPAA, GDPR, FCA, FINRA, etc.)
- Complete content inventory with owners identified for each article
- Governance policy sign-off from legal or compliance teams
Skill and Readiness Checks
Required internal capabilities:
- Content governance owner — assigns a knowledge manager or compliance lead to own the process
- Regulatory familiarity — team members who can interpret HIPAA, GDPR, FCA, or other applicable frameworks
- Escalation path — a defined route for content that needs specialist sign-off before publication
Key Compliance Variables That Shape Your Knowledge Base Governance
Your governance framework isn't one-size-fits-all. How you structure reviews, approvals, and publishing cycles depends on four key compliance variables — and getting them wrong creates real regulatory exposure.
Regulatory Framework and Industry Mandates
The applicable regulation directly determines what content must be reviewed, by whom, and how often. Each framework sets distinct obligations:
- HIPAA (healthcare) — governs PHI handling in clinical and patient support content
- GDPR (data privacy) — controls how PII is referenced or processed across channels
- FCA rules (UK financial services) — mandates fair, clear, and not misleading customer communications
- FINRA (US broker-dealers) — requires review and approval of agent-facing communications
Organizations operating under multiple frameworks face compounded complexity. Misaligned review cycles mean agents may serve outdated content — and that alone can constitute a violation, regardless of whether any harm occurred.
Content Sensitivity Classification
Not all knowledge base content carries the same compliance risk. Content involving PII, financial advice, clinical protocols, or legal disclosures requires more rigorous governance than general product FAQs.
Misclassifying high-risk content as low-risk creates compliance gaps. The reverse — over-governing routine FAQs — creates unnecessary bottlenecks that slow agents down without reducing any real risk.

Channel and Audience for Content Delivery
Governance requirements differ depending on whether the knowledge base serves internal agents only, self-service customers, or both. Content delivered via chatbots or self-service portals often faces stricter disclosure and accuracy obligations than internal agent guides.
Channel-specific requirements matter. Consumer protection rules around self-service financial guidance, for example, apply the moment content is customer-facing — even if your internal governance is otherwise sound.
Rate of Regulatory Change in the Operating Environment
Industries with frequent regulatory updates — financial services, healthcare during a pandemic, telecom amid new data laws — require more agile governance structures. That means shorter review cycles, real-time alerts when regulations change, and faster publishing workflows.
When governance can't keep pace with regulatory change, the gap is immediate: agents work from outdated content, error rates climb, and every interaction carries elevated regulatory risk.
Common Governance Mistakes That Create Compliance Risks in Regulated Knowledge Bases
Most compliance failures trace back to the same handful of avoidable process gaps.
Skipping content ownership assignment: When no named owner exists for a knowledge base article, review cycles stall, updates get delayed, and outdated information persists. It's the most common source of audit failures. HHS required privacy practice changes in 31,191 HIPAA cases — the most frequent resolution pathway — pointing to systemic ownership gaps across affected organizations.
Treating version control as optional: Organizations relying on informal "latest version" conventions cannot demonstrate compliance to regulators or legal teams — even when the content itself is accurate. ISO 27001 Clause 7.5.3 is explicit: previous versions must be archived and rendered inaccessible to prevent use of outdated information. No audit trail means no defensible compliance record.
Applying uniform review cycles to all content regardless of risk: Treating a general FAQ with the same urgency as a clinical protocol wastes resources on low-risk content and under-governs high-risk content simultaneously. Tiered review schedules — quarterly for high-risk, annual for low-risk — are both practical and compliance-aligned.
Failing to update governance policies when regulations change: A framework aligned to last year's regulatory environment creates a false sense of compliance. Governance documentation needs its own scheduled review trigger — especially when applicable laws change. NIST SP 800-53 Rev. 5 requires baseline reviews "when required due to circumstances" and "when system components are installed or upgraded," establishing event-driven governance as a direct regulatory expectation.

Conclusion
Content governance in regulated industries comes down to one thing: accountability. Every piece of information in a knowledge base must be reviewed, approved, and traceable to someone responsible for it.
The most common compliance failures don't stem from bad intentions. They come from poor process design: missing ownership, absent audit trails, and review cycles that aren't tied to regulatory timelines. Getting these structural elements right protects the organization, the agent, and the customer.
The financial case is equally clear. Data breaches involving non-compliance cost an average of $4.61 million — $174,000 more than breaches at compliant organizations. At that scale, a governance framework is a risk mitigation investment, not an operational burden.
Frequently Asked Questions
What are common content governance frameworks (e.g., the 3 C's, 5 C's, and 4 P's)?
Common frameworks include the 3 C's (Content, Context, Channel), the 5 C's (which adds Compliance and Continuity), and the 4 P's (People, Process, Policy, Platform). Regulated industries require at least the 5 C's model to treat compliance as a distinct governance pillar.
What is content governance in regulated industries?
Content governance in regulated industries is a structured system of policies, roles, review cycles, and technology controls ensuring knowledge base content stays accurate, auditable, and compliant with regulations such as HIPAA, GDPR, and SOC 2. Unlike standard editorial governance, it carries direct legal and regulatory accountability.
How often should knowledge base content be reviewed in regulated industries?
Review frequency should be tiered by content risk level. High-risk compliance-sensitive content (legal disclosures, clinical protocols, financial procedures) typically requires quarterly review. General operational content can be reviewed semi-annually or annually. All content should undergo additional review whenever applicable regulations are updated.
What happens if a regulated organization fails a knowledge base compliance audit?
Consequences range from regulatory fines and remediation orders to reputational damage — and in severe cases (healthcare, financial services), suspension of operating licenses. A documented governance process with clear audit trails is the key factor in mitigating penalties, even after a violation has occurred.
What features should a knowledge management platform have for regulated industries?
Look for platforms that offer:
- Version control with full audit trails
- Role-based access controls
- Content expiry alerts and mandatory review workflows
- Compliance certifications (GDPR, SOC 2, ISO 27001, HIPAA)
- CRM and ticketing system integrations to surface governed content at the point of agent interaction
How does content governance differ from content strategy in a regulated knowledge base context?
Content strategy defines what information should be in the knowledge base and why. Content governance defines how that information is created, reviewed, approved, published, and retired. In regulated industries, governance is the compliance enforcement layer that operationalizes the content strategy.


